Oncologists' reflections on patient rights and access to compassionate use drugs: A qualitative interview study from an academic cancer center | PLOS ONE

I'm Willing To Try Anything': Compassionate Use Access To Experimental Drugs And The Misguided Mission Of Right-To-Try Laws | Health Affairs

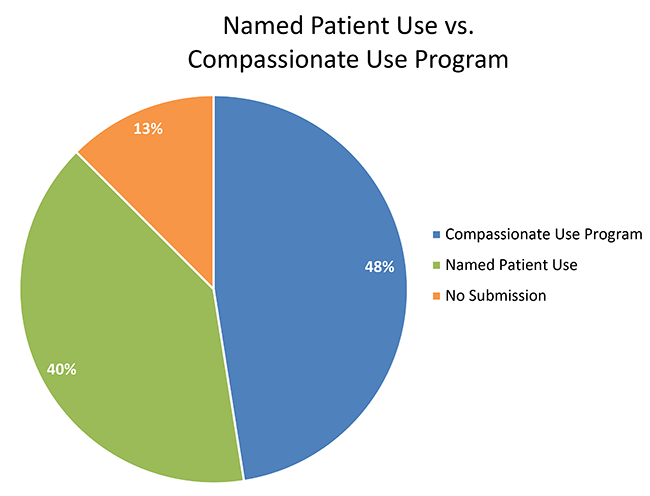
Proportion of Gaucher Patients per country receiving their treatment on... | Download Scientific Diagram

Compassionate use of drugs and medical devices in the United States, the European Union and Japan - ScienceDirect

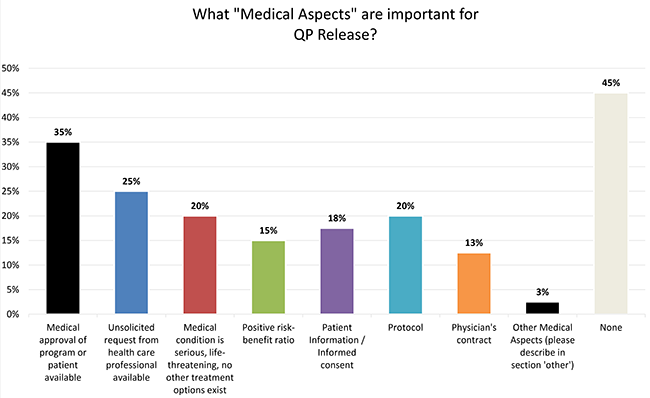
Understanding the challenges and ethical aspects of compassionate use of drugs in emergency situations Goyal PK, Mathur R, Medhi B - Indian J Pharmacol

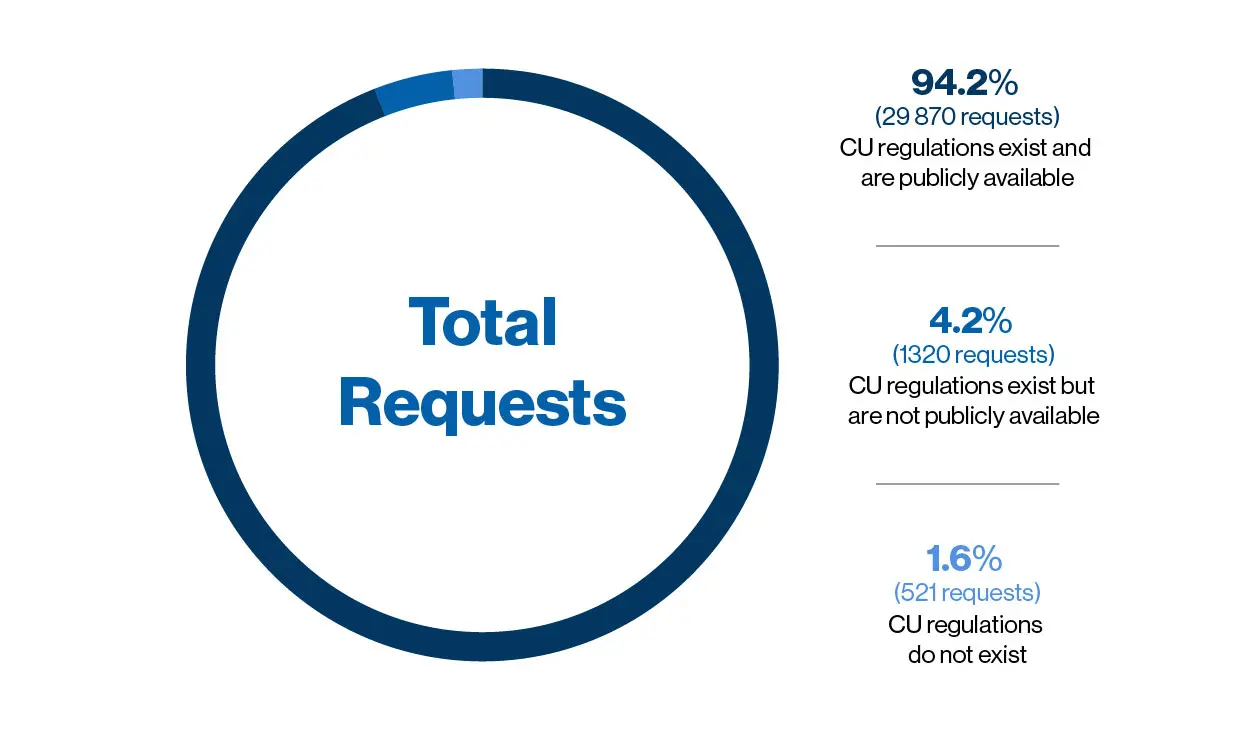
Legal basis, terminologies, and regulatory evidence for compassionate... | Download Scientific Diagram

Compassionate use programs and the European regulatory system Filip Josephson M.D., Ph.D. Clinical Assessor. - ppt download