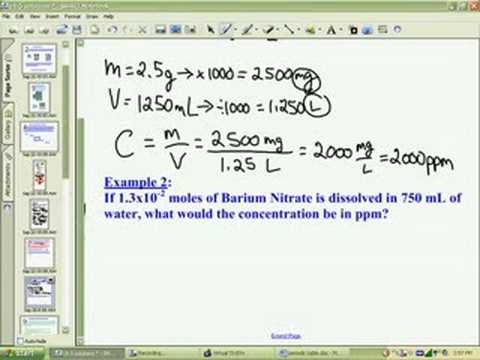
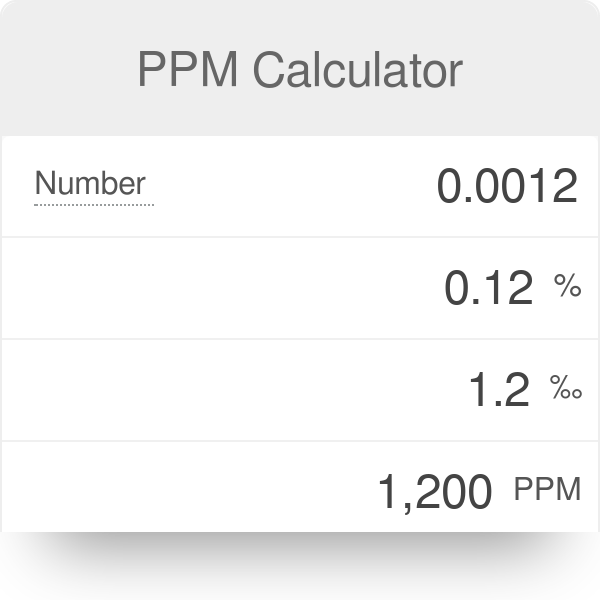
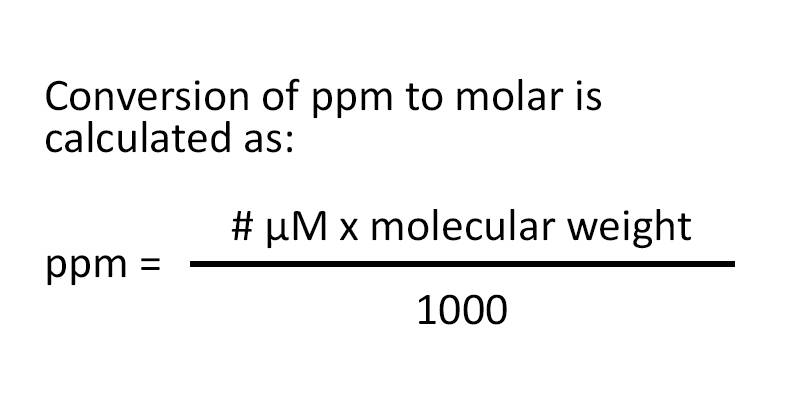
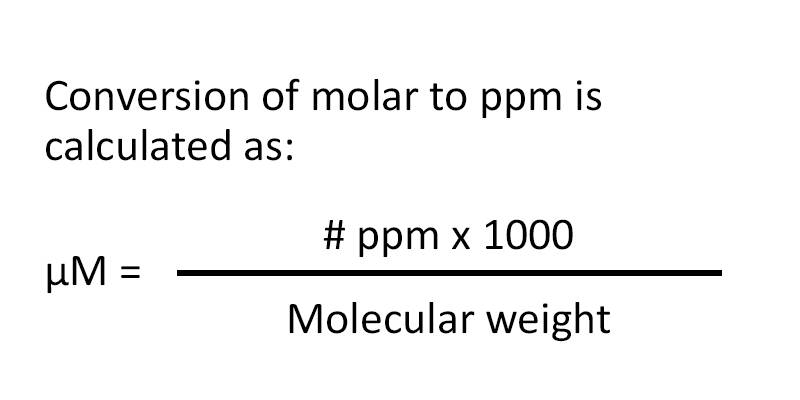
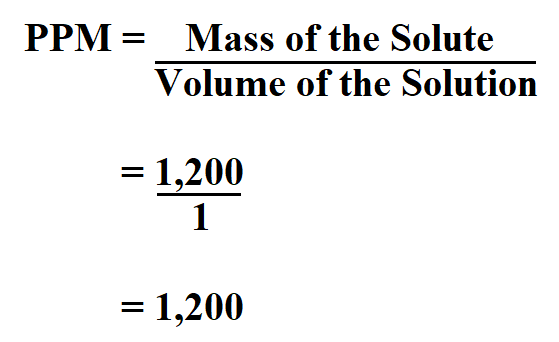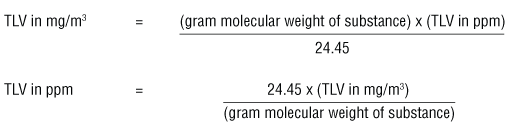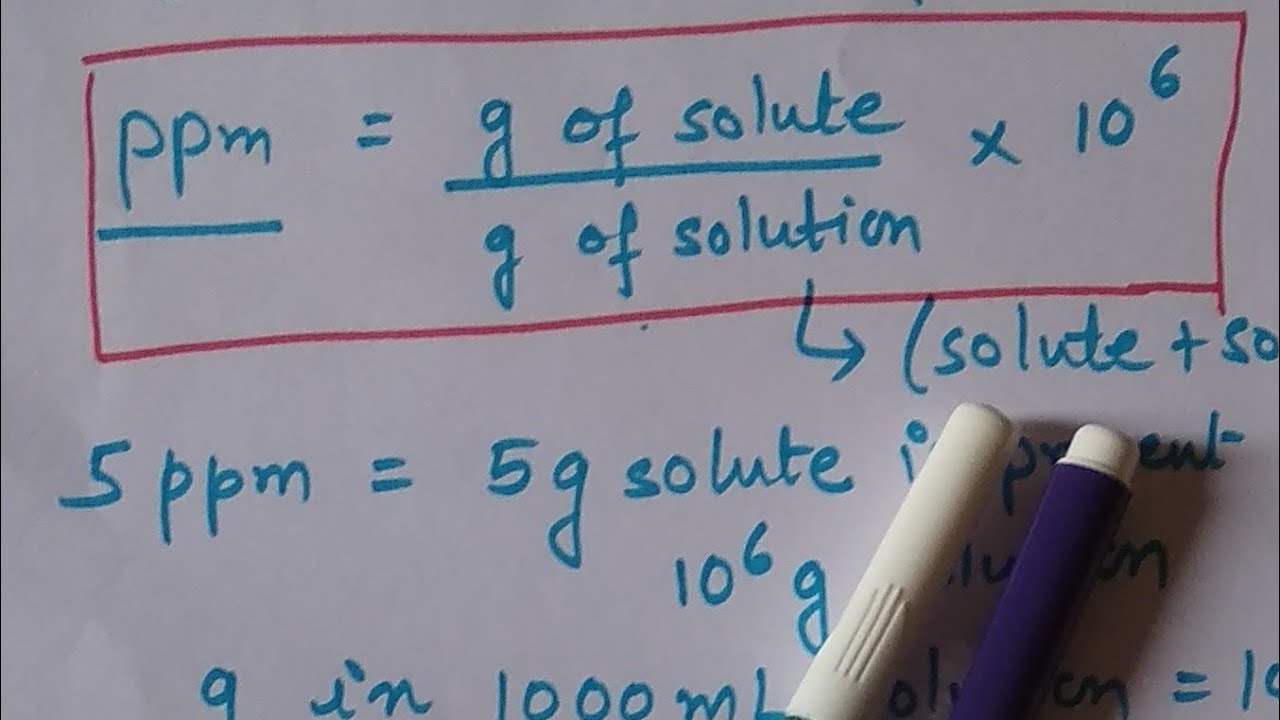White Board Review Practicing with Concentration Expressions Molarity Percent ppm © Mr. D. Scott; CHS. - ppt download

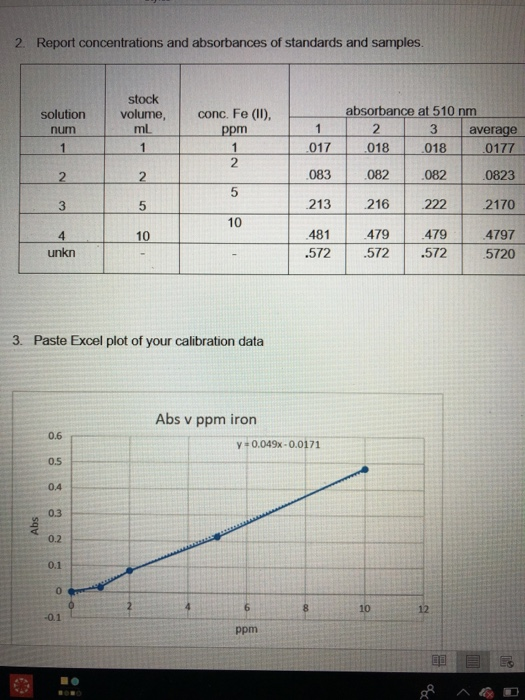
SOLVED: Using the following data of standard Iron solution below, calculate the concentration of an unknown Iron solution. Iron Standard (ppm) absorbance 0.05 0.15 0.10 0.25 0.15 0.34 0.20 0.46 0.25 0.55