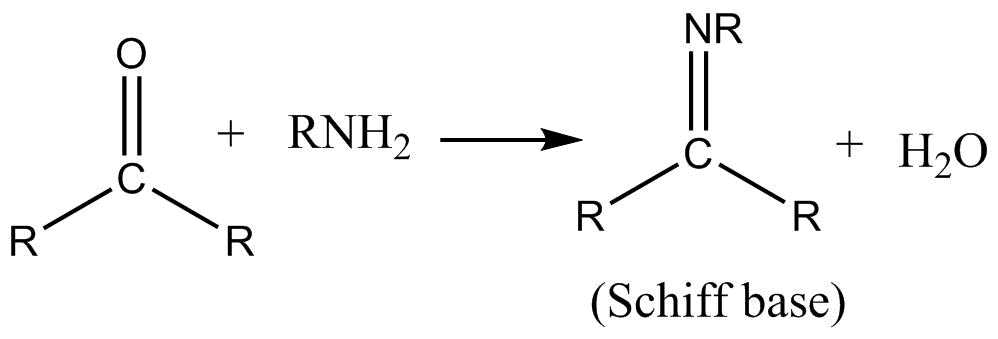
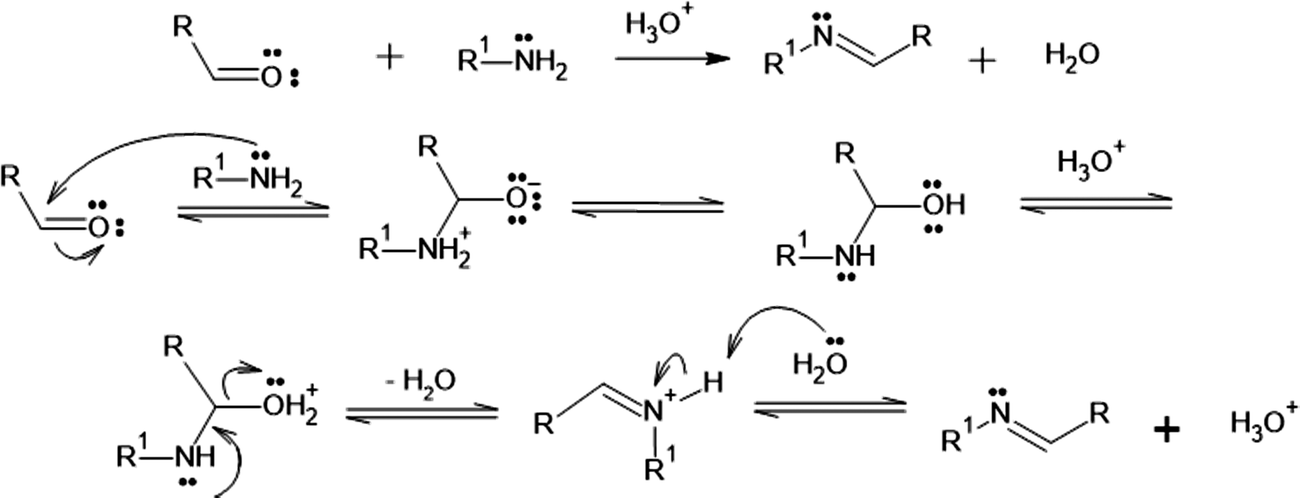
![New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents [PeerJ] New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2020/ochem-4/1/fig-1-full.png)
New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents [PeerJ]

Computational studies on Schiff-base formation: Implications for the catalytic mechanism of porphobilinogen synthase - ScienceDirect

Recent Advances in the Catalytic Applications of Chiral Schiff‐Base Ligands and Metal Complexes in Asymmetric Organic Transformations - De - 2022 - ChemistrySelect - Wiley Online Library

A review on versatile applications of transition metal complexes incorporating Schiff bases - ScienceDirect

Reversible reaction of a Schiff base formed from an aldehydes or ketones. | Download Scientific Diagram

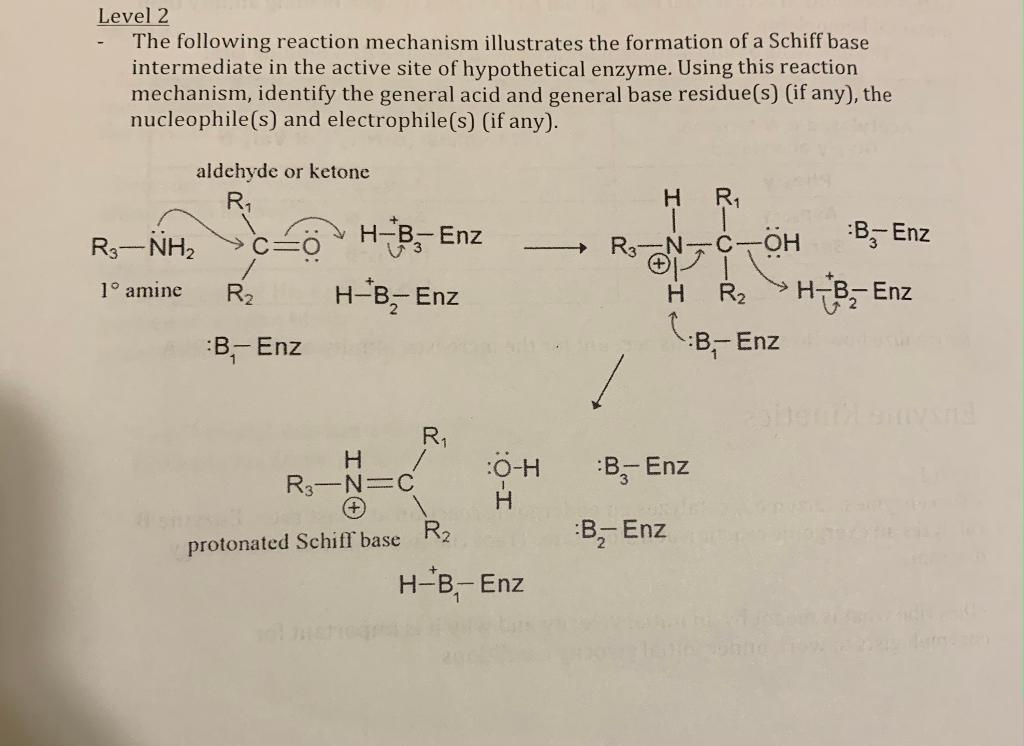
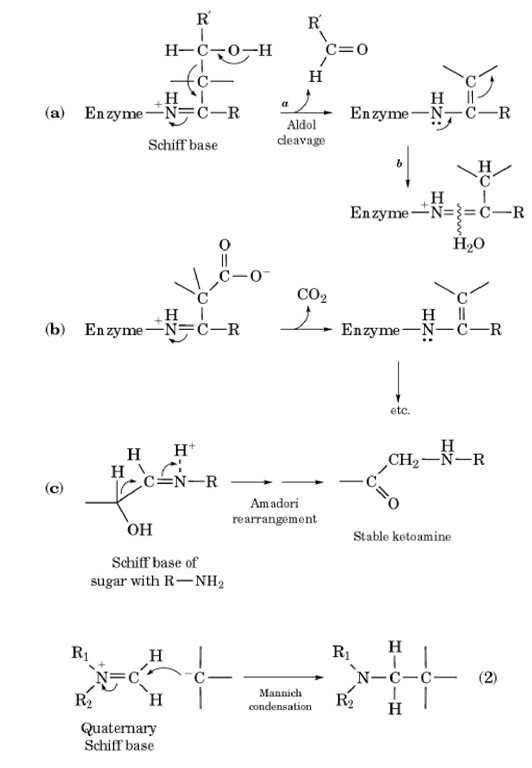
Enzymatic reaction mechanism of type I aldolases Note: After a Schiff... | Download Scientific Diagram
Dynamic protein and polypeptide hydrogels based on Schiff base co-assembly for biomedicine - Journal of Materials Chemistry B (RSC Publishing) DOI:10.1039/D2TB00077F
New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents

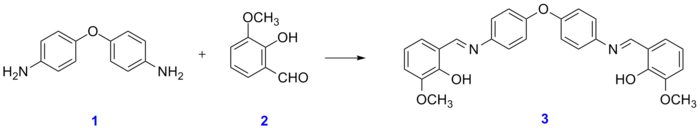
Synthesis, Spectral Characterization and Thermal Behavior of Newly Derived La(III), Co(III) and Mn(II) Complexes with Schiff Base Derived from Methionine and Salicylaldehyde

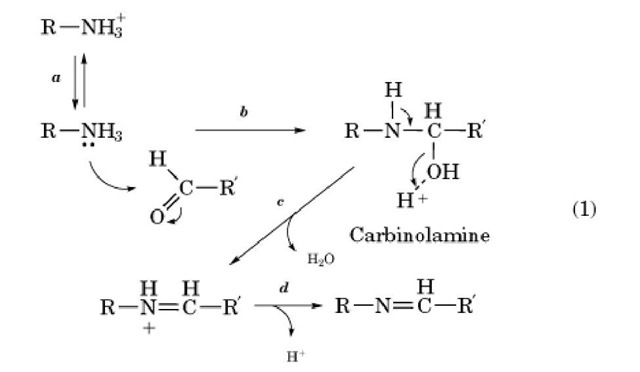
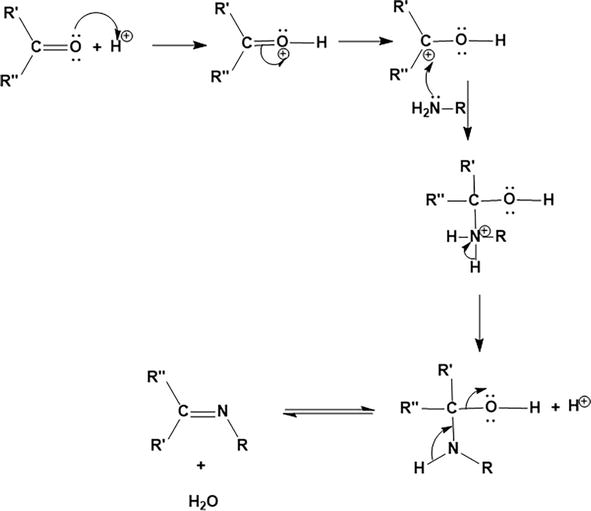
Scheme (1-1): Reaction mechanism for the formation of Schiff bases & OR... | Download Scientific Diagram

Schiff base complexes and their versatile applications as catalysts in oxidation of organic compounds: part I - Al Zoubi - 2017 - Applied Organometallic Chemistry - Wiley Online Library
Schiff Bases and Their Metal Complexes: Synthesis, Structural Characteristics and Applications | IntechOpen