![Acids: taste sour and cause dyes to change color. Bases: taste bitter and feel soapy. Arrhenius: acids increase [H + ] bases increase [OH - ] in solution. - ppt download Acids: taste sour and cause dyes to change color. Bases: taste bitter and feel soapy. Arrhenius: acids increase [H + ] bases increase [OH - ] in solution. - ppt download](https://slideplayer.com/8140591/25/images/slide_1.jpg)
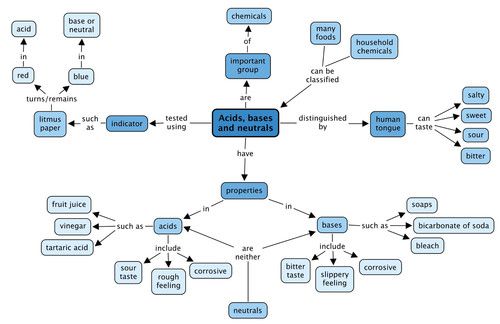
Acids: taste sour and cause dyes to change color. Bases: taste bitter and feel soapy. Arrhenius: acids increase [H + ] bases increase [OH - ] in solution. - ppt download

Acids, Bases, and Salts - Acids taste sour, will change the color of an indicators (chemical dyes), and can be strong or weak electrolytes (aqueous solutions. - ppt download

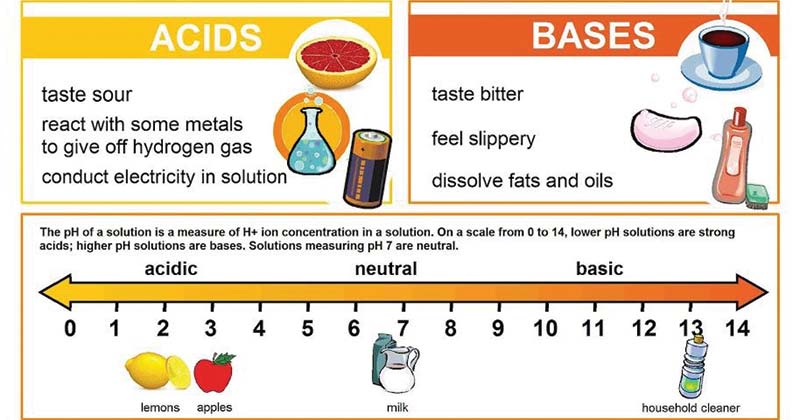
Unit 13 Acids and Bases. A. Properties & Examples electrolyte turn litmus red sour taste slippery feel turn litmus blue bitter taste sticky feel electrolyte. - ppt download

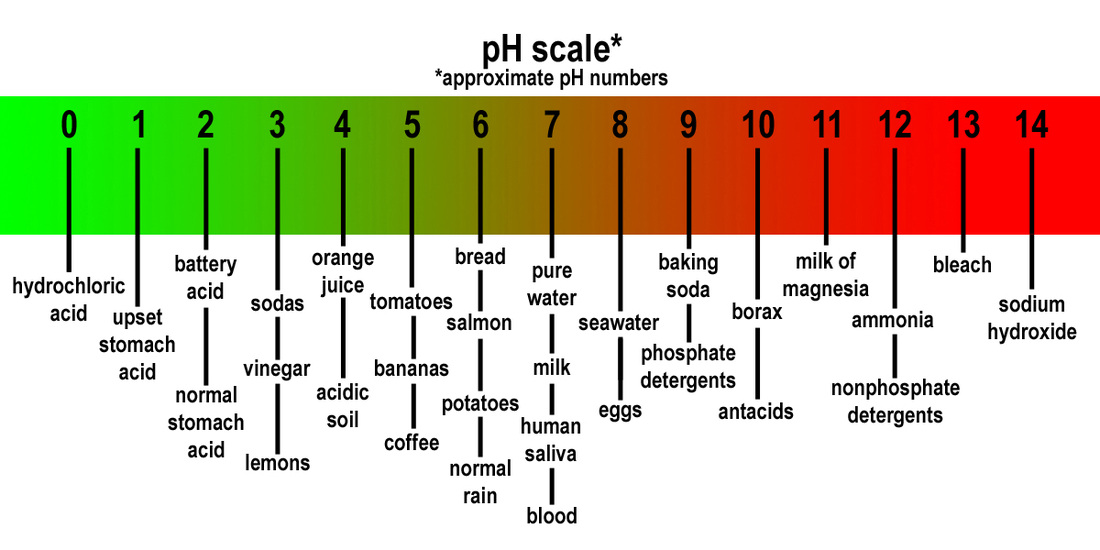
1 4.7 Acids and Bases. Properties of Acids and Bases Acid Base - pH 7 - tastes sour- tastes bitter - no special feel- feels slippery - turns blue litmus. - ppt download

Acid and bases, Identify the following properties as: acid, base or both 1. It turns litmus paper to blue 2. - Brainly.ph

Acids: The Term Acid, In Fact, Comes From The Latin Term Acere, Which Means Acids Taste Sour, Are Corrosive To Metals, Change Litmus (a Dye Ppt Download | le-cerf-volant.ch

PPT - Acid-Base Equilibria: Acids and Bases What makes an Acid an Acid? An acid possess a sour taste PowerPoint Presentation - ID:3209360